What do airports, soda, and Nazi Germany have to with the van der Waals equation of state as applied to carbon dioxide?
Introduction
We will now examine how the van der Waals equation applies to isotherms for carbon dioxide (CO2).
Carbon dioxide is a common gas that is odorless, colorless, and water soluble, and is found mainly in air. It is vital to life, but deadly if we breathe too much of it in. We expire it when we exhale, and plants use it during photosynthesis. CO2 is emitted during the combustion of fossil fuels and also by man-made activities . In solid form, CO2 is dry ice, which is quite handy in refrigeration .
Carbon Dioxide Isothermal Plot
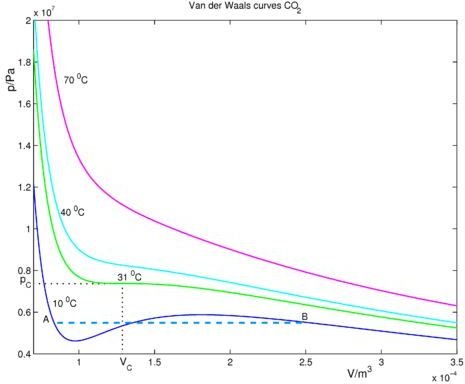
Figure 4, courtesy of Citizendium.org, shows a van der Waals plot for carbon dioxide. Here, the constant a is 0.366 m6 Pa (pascals) while constant b is 42.9 10-6 m3. The critical pressure is 73.7 kPa or 73.7 atm. At 10oC, we are below the critical temperature of 31oC. Point B is where the gas begins condensing or liquefying, and point A is in the area of total liquefaction. Between A and B is where CO2 is in liquid-vapor equilibrium.
Supersaturated and Supercooled Carbon Dioxide

The supersaturated and supercooled forms of CO2 are not specifically pointed out on this plot, but they are between points A and B.
One supersaturated solution of CO2 is carbonated water, a favorite enjoyed by many the world over, especially when it is flavored or added to another drink.
Supercooled CO2 was once used for nefarious purposes. The Nazis injected it into the fallopian tubes of women suffering from traits they deemed genetically undesirable, such as epilepsy, blindness, deafness, or alcoholism. The subsequent scarring rendered the women sterile. Boastful and merciless gynecologist Dr. Carl Clauberg told SS head Himmler that with only a staff of ten people, he could use the substance to sterilize 1,000 women per day.
A more appropriate use of supercooled carbon dioxide is to freeze fog water at airports , so that they turn into ice crystals that drop to the ground.
Conclusion and Caveats
Ideal gas approximations are still mainly used as the basis for statistical physics, because real gases are close to this behavior under many conditions. But for gases of high density and very low temperatures that are close to liquid condensation, the van der Waals equation of state is a very good approximation, though it is not accurate for all gases under all conditions.
Equations for real gases under other conditions are based on the van der Waals model, but modify the potential energy term. For example, the intermolecular forces we considered neglected repulsive forces between molecules. Newtonian physics was used to treat these molecules as hard spheres that bounced back upon collision with non-elastic walls. Other real gas models take repulsion, elasticity, and quantum mechanics into consideration. Perturbation theory and Monte Carlo theory are some of the techniques used to achieve more accuracy for the potential energy term.
Image Credits
van der Waals for CO2 from en.Citizendium.org
Sodas by Austin C. Kiser
References For the Series
Books
Classical and Statistical Thermodynamics by Ashley H. Carter
Physics for Scientists and Engineers by Douglas Giancoli
Statisical Mechanics by Donald A. McQuarrie
Physics For Scientists and Engineers by Raymond Serway
Articles
Nazi Medical Ethics: Ordinary Doctors? by Robert N. Proctor, Ph.D., published in Military Medical Ethics, Volume 2
Resources
Shockwave video by Chemistry Department at Okalahoma State Comparing an Ideal Gas with Real Gases (includes carbon dioxide; click several times on the graph)
Learn How An Internal Combustion Engine Works
This post is part of the series: The Equation of State for Real Gases
A basic introduction to van der Waal’s equation of state for real gases.