The ideal gas law fails for gases that are near condensation. A physics graduate student came up with a modified equation by amending the kinetic theory of gases. How did he do this, and who was he?
Limitations of the Ideal Gas Law
Real gases approximate ideal gas behavior at relatively low densities and atmospheric pressure. But for deviations outside of these ranges, near condensation, the ideal gas law is no longer accurate enough. A more precise representation had to be found. The problem was eventually solved by a physics grad student who later on became a Nobel prize winner.
J. D. van der Waals
The grad student in question was J. D. (Johannes Diderik) van der Waals (1837 - 1923), a Dutch physicis
t. Van der Waals became inspired to study thermodynamics by reading a treatise by Clausius . Despite van der Waals’ being a self-taught school teacher armed with only an elementary education, he was allowed to enter the University of Leyden. A lack of knowledge of classical languages prevented him from enrolling as a regular student to obtain degrees, but he took courses. He was also able to earn teaching certificates in math and physics by studying in his spare time. Afterwards, he became a secondary school director. New regulations finally let science students be exempted from the classical language requirements, and he was allowed to take doctoral qualifying exams, which he passed with flying colors. At the age of 35 and with a wife and kids, van der Waals earned his doctorate in physics. For his dissertation, he derived his eponymous equation of state.
Van der Waals arrived at the equation by considering the kinetic theory of gases , but with some important differences.
Differences From the Basic Postulates
Van der Waals modified the basic postulates of kinetic energy in two significant ways.
Volume: In our previous examination of the basic postulates, we ignored the volume of the molecules themselves, focusing on the total volume of the container. This allowed us to assume that all of the molecules were point masses, and that they were evenly distributed throughout the container. Van der Waals decided that the space or volume occupied by individual gas molecules should be taken into account. Thus the assumption of the uniform distribution of the molecules throughout the entire container no longer holds true.
Intermolecular Forces: These were neglected, as we considered them to be too small compared with the kinetic energy generated by molecular collisions. Only direct contact between the molecules was allowed into our calculations. Van der Waals assumed that these forces are actually quite large, being greater than the size of the molecules themselves, and hence must be taken into consideration. Therefore, there is indeed an action at a distance, albeit quite small and weak.
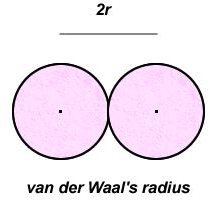
These two changes transformed the basic postulates into something more fitting for real gases, and allowed the derivation of van der Waals’ formula. As before, assume the molecules are spherical with a radius of r, and that they collide with each other but bounce back. These collisions will occur when the distance between their centers become small, as shown in figure 2.
In the next article , we will use the ideal gas law and van der Waals’ postulates to derive the equation of state for a real gas.
Image Credits
Stamp from Physics Related Stamps
Resources
This post is part of the series: The Equation of State for Real Gases
A basic introduction to van der Waal’s equation of state for real gases.


