Four stroke internal combustion (IC) engines using gasoline as the fuel work on the principle of thermodynamic Otto cycle. These are engine are also called Spark Ignition (SI) engines and are employed extensively in the vehicles used by us in our day-to-day life.
Introduction
Thermodynamic Otto cycle is considered to be one of the most commonly used practical cycles. Present days’ all four stroke internal combustion (IC) engines used in your gasoline cars, trucks, motorcycles, constructions machines etc work on the principle of four strokes of Otto cycle. The internal combustion engine working on the principle of Otto cycle is called Spark Ignition (SI) engine in which used gasoline as the fuel. The four strokes of the Otto cycle or four-stroke engine are: suction of fuel, compression of fuel, combustion and expansion of fuel and exhaust of burnt fuel. The four stroke engine has been described in details in the article:
Four stroke thermodynamic Otto cycle was invented by the German scientist Nicolaus August Otto in the year around 1862 when he carried many experiments on heat engine working on four-strokes. He also founded the first engine company, “N.A.Otto & Cie,” along with Eugen Langen. In the year 1867 they won gold medal at Paris Exposition for their contribution to various findings of the four stroke engine.
Otto Cycle
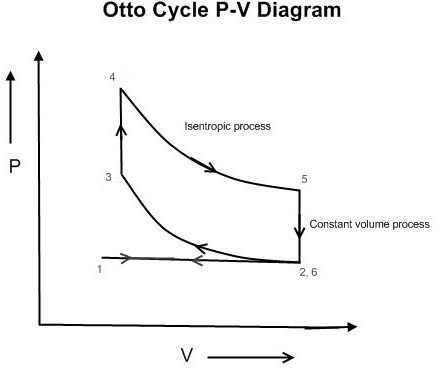
Various Processes of Ideal Otto Cycle
The ideal Otto cycle comprises of two isentropic (constant entropy) and two constant volume processes . The Otto cycle is an open cycle or non-cyclic process since the fresh air-fuel is mixture is inducted inside the engine during each cycle and the burnt mixture is released to the atmosphere. To understand these processes let us consider piston and cylinder engine air-fuel mixture as the working fluid. Refer the P-V diagram given at the top.
Air-fuel intake process 1-2: During this process the inlet valve of the engine is open, the piston moves towards the bottom position inducting air-fuel mixture at constant pressure.
Isentropic compression process 2-3: During this process the inlet and exhaust valves of the engine remain close and the air-fuel mixture, which has been inducted inside the cylinder, is compressed to the minimum volume.
Combustion of air-fuel mixture at constant volume 3-4: Thereafter, the air-fuel mixture inducted inside the cylinder is combusted by the spark at constant volume; hence these engines are called spark ignition engines. This leads increase in temperature and pressure inside the cylinder.
Isentropic expansion process 4-5: Due to extremely high pressure, the piston is pushed again towards the bottommost position of the cylinder. It is during this process that the actual work is produced from the engine.
Constant volume process 5-6: During this process the exhaust valve opens and all the exhaust gases are ready to be released to the atmosphere. The pressure inside the cylinder falls drastically.
Exhaust process 6-1: During this process the exhaust valve is open and the piston moves upwards and removes all the exhaust gases inside the cylinder at constant pressure.
Thereafter the exhaust valve closes, the piston starts moving in downward direction, the inlet valve opens and fresh air-fuel mixture is inducted. The whole cycle is completed in four strokes of engine, hence it is called four-stroke engine.
Efficiency of Otto Cycle
The efficiency of Otto cycle is given as: 1- (T4-T1)/(T3-T2)
Where T1, T2, T3, T4 are the absolute temperatures measured in degree Kelvin
The Otto cycle was an important step in the history of thermodynamics and vital step in the progress of the human beings. All the previously invented engines like Carnot , Stirling , Ericsson and other cycles related to heat engines were more of for the theoretical studies of the engines, but Otto cycle laid important basis for the practical engines, which went to becomes the most widely used engines and marked a great leap in the human progress for the centuries to come.
Next: Part-3 of this article
Previous: Part-1 of this article
Reference
Book: Engineering Thermodynamics by P K Nag
Related Readings
Thermodynamic Stirling Cycle and Stirling Engine - Part 1
Carnot Cycle and Carnot Theorem: Working and Relation to Second Law of Thermodynamics – Part 1
Types of Thermodynamic Systems and Important Terms Related to Thermodynamics - Part 1