Thermodynamic Stirling cycle comprises to two reversible isothermal and two reversible isochoric processes. In the initial years the Stirling engines were used frequently but were later replaced by other engines. In the recent years they have been gaining their lost ground.
Introduction
The Stirling cycle is the thermodynamic cycle on which the Stirling engines work. It was invented by Scottish Church Minister, Robert Stirling with the help of his brother, who was an engineer, in the year 1817. He further developed the engine and got it patented. Stirling cycle was invented before Carnot cycle which was first discussed in the book, “Reflections on the Motive Power of Fire,” in the year1824 by young French scientist, Sadi Carnot.
In Stirling cycle gas like air, helium, hydrogen etc is used as the working fluid. All the processes of Stirling cycle are assumed to be reversible process, hence when the gas is heated the engine produces work or power and when work is supplied to the cycle it works as the refrigerator or the heat pump. The reversed Stirling cycle is used extensively in the field of cryogenics as the cryogenerator to produce extremely low temperatures or liquefy gases like helium and hydrogen.
Processes of Ideal Stirling Cycle
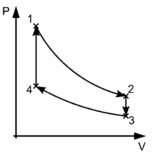
The ideal Stirling cycle comprises of two isothermal processes and two constant volume processes. The rejection of heat and absorption of heat takes place at constant temperature. Here are various processes of the Stirling cycle; these have been described with the piston-cylinder diagram and P-V diagram.
- Isothermal expansion process 1-2: During this process the piston starts moving towards the outer part of the cylinder hence the pressure of the gas reduces and its volume increases. This is the part of the cycle when the work is produced by the cycle. The gas is heated at the constant temperature or isothermally at temperature T1.
-
Constant volume process or isochoric heat removal process 2-3: During this process heat within the fluid or gas is transferred to the regenerator for the use in next part of the cycle. The temperature of the gas reduces to T3.
-
Isothermal compression process 3-4: During this process the piston starts moving inside the cylinder hence compression of the gas occurs at constant temperature. Whatever the heat is generated during this process is rejected to the coolant in the intercooler.
-
Constant volume process or isochoric heat addition process 4-1: During this process the compressed gas moves towards the regenerator and absorbs the heat and its temperature increases to T1.
Thereafter the gas expands isothermally producing the work as described in process 1-2. In this way the cycle keeps on repeating.
Stirling Cycle P-V Diagram (Source: Wikipedia)
Efficiency of the Stirling Cycle
The efficiency of Stirling cycle is given as (T1 – T3)/T1 or 1-T3/T1
Where T1 and T3 are absolute temperatures measure in degree Kelvin
Next: Part-2 of this article
References
Related Articles
Carnot Cycle and Carnot Theorem: Working and Relation to Second Law of Thermodynamics – Part 1
Types of Thermodynamic Systems and Important Terms Related to Thermodynamics - Part 1