Learn about the main battery types that are used on board a ship including their internal working details with the aid of diagrams.
Introduction
Batteries can be an excellent tool for storing electricity and can also be used as a alternate source for power. Used on almost all the ships, batteries are often used as instantly available energy supply source. However, batteries can also be used on a regular basis to provide a low-voltage dc supply to many engine and deck machines. The size and type of the batteries to be selected generally depends on the application and capacity of the services.
Usually, two main types of batteries are used on board:
- Lead- Acid Battery
- Alkaline battery
In this article we will take a look at how do lead acid and alkaline batteries work and internal battery construction details in the subsequent sections
Lead- Acid Battery
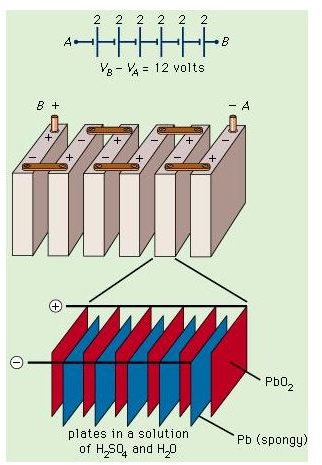
The Lead – Acid Battery used on board a ship consists of a series of cells, with each cell containing a lead peroxide positive plate and a lead negative plate immersed in a dilute sulphuric acid solution. This sulphuric acid solution is known as electrolyte. The whole arrangement is kept in a leak-proof casing. A wire joins the two plates. This develops a potential across the wire and a current starts flowing through it. The voltage thus developed has a measure of around 2.2 volts initially and a steady value of 2 volts. All the Lead Acid batteries used on board generally have six separate cells connected in series, which means that the total power output each battery develops is around 12 volts. These batteries are also known as accumulators.
Modern Lead Acid Batteries uses many interleaved plates in a single cell to produce a compact arrangement with greater capacity. The whole arrangement, in order to create proper insulation, is surrounded by heavy duty plastic, hard rubber or bitumen case.
The adjacent picture clearly shows the internal nitty gritty of such a lead acid battery which consists of 6 cells of 2 Volts each connected in series which gives rise to a total output voltage of 12V. The circuit diagram at the top shows the symbolic representation while the middle part of the picture shows the physical arrangement. Finally the bottom most portion of the image shows how each individual cell is further subdivided into the electrodes as explained above.
Alkaline battery
The alkaline battery used on board a ship consists of a nickel hydroxide positive plate and a cadmium and iron negative plate. Both the plates are immersed in a potassium hydroxide solution to produce a voltage of about 1.4 volts. A usual battery used on board consists of 5 cells which produces a total of 7 volts.
The modern alkaline battery consists of many interleaved plates for higher capacity. The whole arrangement is mounted in hardwood crates with spaces between each cell. All the cells are connected in series and the casing itself is considered “live” because of its contact with the plates and electrolyte.
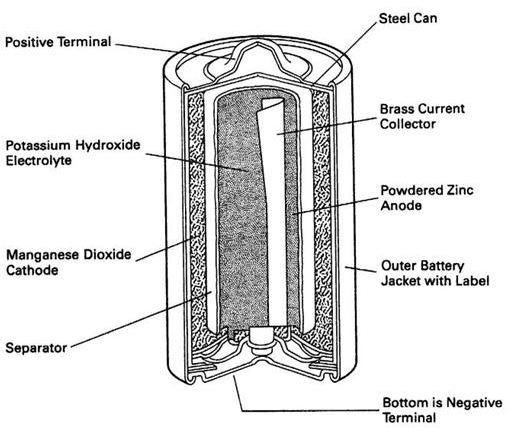
The full constructional details as seen in the adjacent diagram are self explanatory and each and every part is clearly labeled. The reader is advised to go through the diagram carefully to fully understand the working concept of such batteries
Image Credits
RadioShack Corporation 1990
Britannica.com