pH of water is an indicator of its acidity or alkalinity and ideal value of drinking water pH is 7. Just find out how this parameter is measured on board ships
pH of SeaWater
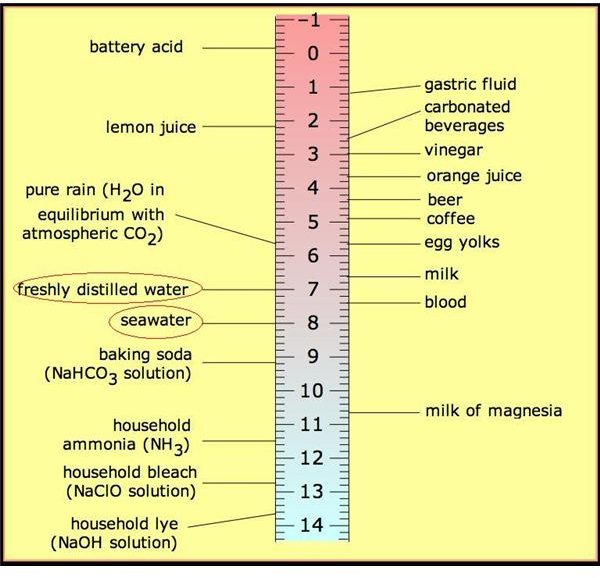
The pH value of any solution gives an indication of the solution either being acidic, alkaline or neutral. The scale of pH varies from 0 to 14 with 7 as the midpoint. A neutral solution has the pH value of 7 and anything less than that is acidic in nature while above 7 indicates alkaline solution.
The image above shows the pH scale and the pH values of solutions of commonly found substances. As you can see the pH of sea water is slightly alkaline in nature while that of fresh drinking water is (at least ideally) neutral. This is obvious since you do not want to be drinking acid or base. Hence it is very important to measure the pH of water before consumption on a ship apart from measuring its salinity and ionic purity. Apart from that it is even more important to measure pH of water used for boiler feed pumps and engine cooling systems of both the main engine and auxiliary engines as any significant change in the pH from neutral value can cause extensive damage through corrosion.
pH Meter
As you might know there is a very simple method of measuring pH by using pH strips which simply involves dipping the strip in the solution and noting the change in its colour and comparing the same with a chart which is supplied along with the strip packet. This is a very easy, economical method to measure pH but only used for checking at intermittent intervals. When dealing with a situation such as the pH of drinking water on a ship, we can certainly use this method by checking the pH of water storage tanks regularly. But other situations such as feed water systems require continuous monitoring of pH so that there is no risk of the water being acidic or alkaline at any time.
The setup used to measure continuous pH is shown in the diagram below which shows a typical pH meter used on board ships. It consists of two electrodes out of which one acts as the reference electrode while the other is the measuring electrode. The meter measures the concentration of hydrogen ions which are actually dissociated in water and give an indication of the pH value of the solution under test as shown in the sketch.
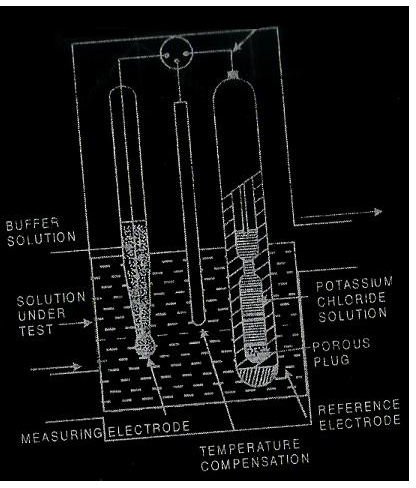
The concentration of hydrogen ions is in turn is indicated by the emf developed across a thin membrane of special type of glass and the reference electrode has a constant emf while the measuring electrodes generates an emf which is proportional to the difference between the pH of the solution under test and a solution having a known fixed pH value.
Needless to say there are several factors which could effect the reading such as temperature of the solution (hence a temperature compensation system is provided), electric fields, dirty electrodes and so forth, hence care should be taken to take all these factors into account.