The use of nanoparticles and nanomaterials in fuel cells has provided a hope for a greener environment. However, there are potential risks that should be taken into account if we want to make accurate assessments for the future.
Introduction
The development of fuel cells has given the energy conversion industry a good potential for a greener future. Nanotechnology on the other hand, has promoted fuel cell technology to a higher level, where fuel cells are more efficient, inexpensive, and suitable for a wider variety of applications than before. At this point, it is crucial to examine the enviromental effects of these promising, yet potentially risky advances.
Nanotech Related to Fuel Cells
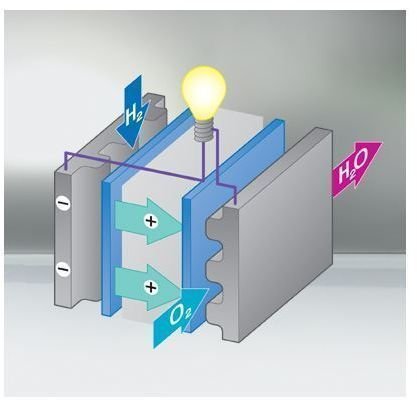
Fuel Cells are electrochemical systems that have the ability to convert chemical energy (burn fuel) in order to generate electricity. For example, a hydrogen fuel cell uses hydrogen as a fuel to produce electric current, as depicted in the image below. In order to assist the chemical reactions taking place inside the cell, catalysts such as platinum are also used. Although platinum is a chemically inert material in its bulk state, it tends to become very reactive in its nano-powder form. So, in order to reduce the quantity of this expensive material without reducing the reactive surface area, companies resorted to the use of platinum nanoparticles, and thus managed to cut down the cost significantly.
Fuel Cells also use selective membranes so that only fuel atoms can penetrate and enter the cell. The use of nanomaterials makes these membranes more efficient and also serves in the development of light and inexpensive fuel tanks especially for hydrogen storage. So the main concern focuses on the environmental effects of nanoparticles and nanomaterials used in fuel cell manufacturing and operation.
A Schematic View of Hydrogen Fuel Cell Operation
How Nanotechnology in Fuel Cells Affects the Environment
The positive contribution of Nanotechnology in fuel cell technology is undeniable. The effectiveness of energy conversion with minimum losses and the reduction of production cost give hopes for a greener environment. But although much is said about the positive effects, even less is known about the potential environmental risks due to extensive nanomaterial and nanoparticle use. All concerns are mostly associated with nanoparticles escaping into the environment. Nanoparticles that are embedded and enclosed inside the cells cannot come into human contact except in the case that they somehow escape from the cell (through being damaged or burned). In addition to that, there’s always the risk of leakage during the production, operation, and disposal of fuel cells.
But why is this leakage so dangerous? Due to the nanoparticles’ high surface reactivity, new chemical reactions could be ignited on the ground or in the air. Their ability to accumulate in the soil and water could interfere with the delicate food chain balance and have unknown effects on ecosystems.
The health impacts of ultrafine nanoparticles to all living organisms could be severe as well. Some of them are toxic when inhaled and could cause great damage to the respiratory and cardiovascular system of humans and animals. On the other hand, their controllable release into the environment could have a positive view as well. Their high surface area could help in the adsorption of molecular contaminants and clear the environment according to a more ambitious plan. Another major issue is the disposal and recycling of the nanoparticles/nanomaterials used in fuel cells. Many new methods are being tested, but they are only in an experimental stage. In order for recycling to become effective, it should move to an industrial scale.
Is it Harmful for the Environment, After All?
The environmental impacts discussed above have only recently started to puzzle the scientific community. The existing information is not sufficient to assess the environmental risks. Apart from that, the assessment should probably apply specifically to each material rather than to the nanostructured form of the materials in general. Since no reliable analysis method exists on this matter yet, we can only speculate for the future.
Sources:
“Opportunities & Risks of Nanotechnologies”, Allianz Center for Technology
“Nanotechnology and the environment: A European perspective”, Science & Technology of Advanced Materials, D.G.Rickerby, M. Morrison (2007)
Nanotechnology in Fuel Cells : “How Can Nanotechnology Improve Fuel Cells?”
Physorg.com: “Recycling nanoparticles”