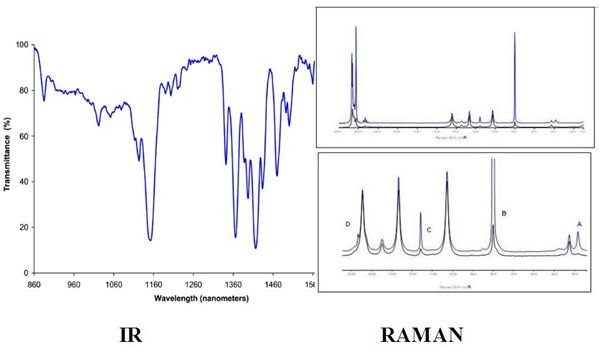
IR and Raman are two similar spectroscopic techniques. However, their comparison has revealed at least four differences that are described below.
Introduction
It is already known that photons interact with molecules and induce transitions among the available energy levels. These transitions result in the emission of photons with various wavelengths. In the case of transitions between vibrational energy levels of molecules, the photons emitted are detected with two spectroscopic techniques, the Infra-red (IR) and Raman Spectroscopy.
Raman and Infrared (IR) Spectroscopy, In a Few Words
When light (or a photon) interacts with a molecule, it can either be elastically or inelastically scattered. In the first case, the absorbed photon has the same wavelength with the emitted one (Rayleigh scattering), but in the second case, the photon has lost or gained an amount of energy related to the vibrational energy spacing in the molecule. This amount corresponds to the wavelength of the Stokes and anti-Stokes lines which are equally displaced from the Rayleigh line in the Raman spectrum. Infra-red (IR) spectroscopy also measures the vibrational energies of molecules. The absorption of light in the infrared region of the electromagnetic spectrum, gives a spectrum that corresponds to specific vibrational modes and is unique to each molecular structure examined.
What’s Their Difference?
The main difference between the two techniques lies in the nature of the molecular transitions taking place. For a transition to be Raman active there must be a change in the polarizability of the molecule during the vibration. This means that the electron cloud of the molecule must undergo positional change. On the other hand, for an IR detectable transition, the molecule must undergo dipole moment change during vibration. So, when a molecule is symmetrical, e.g. O2, we cannot observe any IR absorption lines, since the molecule cannot change its dipole moment. It has been observed that molecules with a strong dipole moment are typically hard to polarize.
Regarding the excitation wavelength, the Raman technique uses a monochromatic beam or laser, in the visible, near-infrared, or near ultraviolet range of the electromagnetic spectrum. In IR spectroscopy, a monochromatic beam is used in the infrared region of the electromagnetic spectrum. The wavelength here increases or decreases over time, in order to observe all the absorption lines within a specific range of the infrared region.
Another difference can be observed in the resulting spectra. The IR technique shows irregular absorbance (or transmittance) lines, depending on the material investigated. The Raman spectrum mainly comprises the elastic scattered light line (Rayleigh) and two equally distanced lines Stokes and anti-Stokes, with the second being rather weak and difficult to detect.
Finally, the Raman technique requires high-stability laser sources and sensitive amplification equipment to detect the weak signal. This makes Raman instruments more expensive compared to the IR equipment. However, the new advances in technology have altered this to some degree.
In Overall
Despite their differences, both techniques serve the purpose of identifying unknown substances. Molecules that cannot be detected with the one method can be easily detected with the other, and that leads researchers to use them both in a complementary way.
References:
“IR and Raman Spectroscopy: Fundamental Processing”, S.Wartewing, 2003
Picking the Right Analysis Method , Laboratory Equipment.
“Infrared Characterization for Microelectronics”, W.S.Lau, 1999
Introduction to Raman Spectroscopy , Horiba Ltd