The article describes the properties of azeotropic mixtures, the distillation process, and the advantages of azeotropes when used as solvents by the manufacturing industry.
What is Distillation?
Before explaining what an azeotrope is, it would be useful to define the process of distillation.
The process of distillation relies on the fact that the different components of a mixture will boil and therefore vaporize at different temperatures.
This is possible because the various ingredients of the mixture have different boiling points. During distillation, a liquid mixture is boiled at a certain temperature and a component of the mixture is vaporized and collected. The rest of the mixture remains in a liquid state. Following this procedure one or more components can be separated from the rest and then cooled and condensed. When applying the distillation process to sea water, the water will evaporate as soon as the temperature reaches its boiling point. The residue will mostly consist of salt, and the condensed vapor is nothing but pure water.
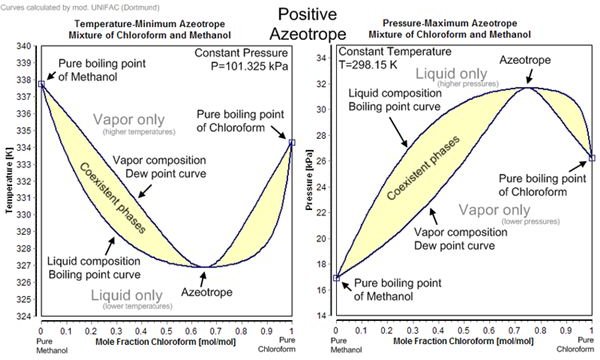
Azeotropes
The most general definition of an azeotrope is to describe it as a constant boiling blend of two or more components. What makes azeotropes different to other mixtures is their ability to keep their composition constant during simple distillation. This means that the vapor of the azeotropic mixture has the same ratio of components as the original mixture.
For example, when an azotrope containing 97% HFC and 3% alcohol is boiled, the vapor will also consist of 97% HFC and 3% alcohol. If we re-liquefy these fumes, the resulting mixture will also have the same ratio of HFC and alcohol as the original. Therefore, the distillation process cannot separate the constituents of this azeotropic mixture.
The preparation of an azeotropic mixture depends on the composition, ratio of components, boiling points, and surface tension of the blend.
Types and Examples of Azeotropes
Depending on the boiling point of the mixture:
Positive azeotropes (or minimum boiling mixture or pressure maximum azeotropes): The boiling point of this azeotrope is lower compared to the boiling points of its components. A well-known example is the azeotrope of ethanol 95.63% with water that boils at 78.2 ºC which is lower than that of water (100 ºC) or ethanol (78.4 ºC) by itself.
Negative azeotropes (or maximum boiling mixture or pressure minimum azeotropes): The boiling point of this azeotrope is higher compared to the boiling points of its components. For example, the mixture of nitric acid (68%) with water that boils at 120.5°C, although nitric acid boils at 83°C.
Depending on the number of components:
- Binary: contains two components
- Tenary: contains three components
- There are also azeotropes that consist of more than three components.
Advantages and Use of Azeotropes in Manufacturing Technologies
Azeotropes are commonly used in industry as solvents. Some of their advantages in this direction are the following:
- One of the most important properties of an azeotrope is the ability to mix flammable and nonflammable constituents to create a nonflammable azeotropic blend. The chemicals of the azeotrope can never be separated when in a hot environment and thus dangerous flammable vapors cannot exist. This is applicable in the production of nonflammable solvents that are safer and more reliable to use especially in industrial facilities.
- Azeotropic solvents can be very effective and fast-performing cleaners.
- They are very ease to handle since they don’t require the mixing of a solvating agent and a rinse agent. A single azeotropic solvent eliminates the need to monitor the ratio of solvating agent and rinse agent, so no mistakes can be made
- The distillation and recovery are very simple processes for the azeotropic mixtures.
- They present desirable properties: they are very mild cleaners with minimal odor that improves functionality within closed spaces.
- They can operate under low temperatures as well and they are very flexible since their operation does not depend on the mixing ratio between the solvating agent and the rinse agent.
- Azeotrope fumes can be reboiled and recycled.
- The cost of azeotropes is considerably lower compared to the costs of two sol-vents used in a co-solvent process. The quantity of azeotrope to use is also lower due to the fact that no loss occurs during the co-solvent process.
Sources:
- www.microcare.com
- Encyclopedia of Chemical Technology, by J. Wiley & Sons, 2007
- www.vertrelsolvents.com
Image Credits:


