Have you ever thought why the sea water is salty and fresh water not? What is it that makes the sea water salty? There is more than one reason that leads to the salinity present in the sea water. Find them out in the article inside.
A Pinch Of Salt
Everyone who has been to a beach for swimming in sea water and has experienced a mouthful of sea water , knowingly or unknowingly, is cognizant of the fact that sea water is salty. The experience must have also left you with an uncanny look and a question pounding in your brain as to why the sea water is salty? Also, why fresh water found in rivers or lakes is not salty?
It is a known fact that fresh water as contains some dissolved chemicals and salts but in very less quantity. The salt concentration of sea water is way too high than that present in the fresh water and that’s the reason for difference in taste. There are bodies of water such as the Dead Sea where salt concentrations are even higher.
There are many reasons that have lead to the saltiness present in the sea. In this article we will take in account, all the congruous and viable explanations that have led to the salinity in the sea.
Reason 1 - Rain Water To Salt Water
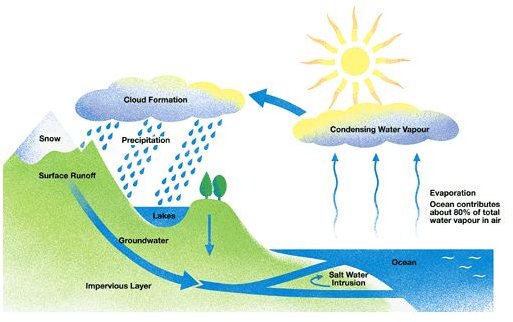
The ocean waters were not salty from the start. There are various reasons, which took place for millions of years before the sea turned salty. One reason states that fresh water is being turned to salty water as a result of various factors and that has led to the present condition of sea. Rain water during its passage through the atmosphere absorb many salts, chemicals and gases. Due to the presence of gases such as carbon dioxide, rain water becomes a acidic in nature. When the rain water falls on the rocks, the acid errodes the rocks and carry them with it in the form of ions. This rain water is carried to the oceans through rivers or streams and gets accumulated there. Millions of years needs to pass before these ions make a significant contribution to the salinity of sea. The process by which rain water permeates through the soil and absorb salts and minerals is known as weathering.
In addition to this, many of the rocks also contain basic salt, i.e Sodium chloride in large quantities. When rivers or rain water flows over them, it carries a significant amount of salt from such rocks which later gets accumulated in the sea.
Reason 2 - Hydrothermal Vents
Apart from the previous reason, there are ridges on the ocean bed that also contribute to the salinity. Some of the rain water seeps deep inside the earths crust. The ocean water itself also seeps through these ridges into the rocks of oceanic crusts. This water gets heated due to the high temperature core of the earth and flows back through these crests into the sea. Along with the hot water comes lots of minerals and salts absorbed from the rocks. Its been predicted that a large amount of hydrothermal fluid flows through these oceanic vents and thus has a substantial contribution to the sea water salinity.
In addition to that, sea water also reacts with the oceanic rocks. Water reacts with rocks to increase the concentration of dissolved salts. Thus there is a continuous production of salts and minerals taking place at the bottom of the ocean.


Reason 3 - Submarine Volcanoes
Just like the volcanos erupting on the surface of the earth, there are underwater volcanic eruptions that takes place on the ocean bed. A large amount of hot lava along with rocks and minerals that erupts out of the surface, reacts with the sea water and produces dissolved salts and minerals.Thus increasing its concentration and thus the salinity.
It’s being predicted that such processes are taking place at such high frequency on the ocean beds that they compensate the amount of salts that are lost in the formation of new minerals.

Summary
The above mentioned, are the main reasons that contribute to the salinity of the sea water. In addition. it’s also predicted that, water cycle has also contributed in a significant way towards increasing the salinity. Oceans spread over a large area on the earth and thus evaporation of water takes place at a very high rate from the oceans. When the water evaporates it leaves behind the dissolved salts. The evaporated water comes back to the oceans in the form of rain, which seeps through the earth’s crust or react with the rocks to make more salt ions. This process must have taken millions of years to make the concentrated salt left behind after evaporation, count in a substantial way.
Image Credits
https://www.north-sulawesi.org/images/mahangetang.jpg
https://farm1.static.flickr.com/152/419595167 _5fa104407b.jpg
https://1.bp.blogspot.com/ _7zvcyyNFXOY/ScbM3s3mPGI/AAAAAAAAC3w/PR6zPBGuy44/s400/Th-eruption-of-underwater-volcanoes002.jpg
https://www.sawater.com.au/NR/rdonlyres/657AC917-D6E3-4E55-AAD1-38119A0ACBB4/0/diag _water_cycle.gif