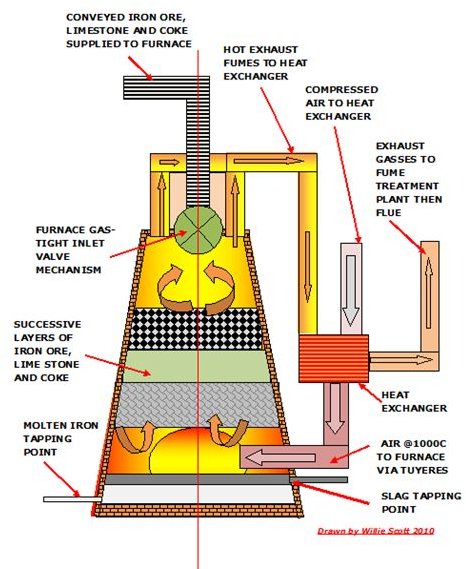
Large scale iron ore smelting has been carried out in numerous types of blast furnaces since the industrial revolution, with the basic ingredients of coke, iron ore and limestone remaining unchanged, being loaded into the blast furnace from where the molten iron is tapped into molds.
Introduction to Iron Ore and Steel Smelting Processing
Iron ore is obtained in the conventional method of open cast or underground mining and conveying the ore to the surface preparation are where it is crushed, washed, and transported to the smelter.
Here the ore is put into a blast furnace along with limestone and coke and subjected to hot air blasting and heat which converts the ore to molten iron. This is tapped from the bottom of the furnace into molds known as pigs and allowed to solidify into pig iron.
This pig iron is converted to wrought iron or processed into steel by several methods.
This is the first article in a series on metallurgy, where we will examine the processing of iron ore into different grades of steel. We start with the mining and preparation of the iron ore and shipping it to the smelter.
The Mining of Iron Ore
Iron ore mining was a thriving industry in many parts of the world, with the smelters being located as close to the mines as possible. However as larger mines where more efficient mining of higher iron content ore became effective, the smaller mines fell by the wayside.
The top four mines with the highest output of iron ore are in China, Australia, the US, and South Africa.
There are two distinct categories of iron ore mining, surface mining such as open cast or strip mining and underground mining.
At the surface iron ore processing plants, the iron ore is put through crushers and ball mills to break it into a uniform and more workable size and then washed with the residue going to tailings.
The ore is the transported to the various smelters located locally or on the other side of the world with large bulk carriers being used to ship the ore by sea.
Iron Ore Smelting Process Using Modern Blast Furnaces.
Although the basic ingredients of an iron ore blast furnace has changed little over the last hundred years or so, the furnaces themselves have been modernized to improve efficiency and the purity of the iron and steel.
The furnace has a more efficient shape to suit automation, with the contents of iron ore, limestone and coke being carefully measured, conveyed and loaded into the furnace from the top through specially designed gas-tight valves. High temperature air at over 1000C (which is can be heated using the exhaust gasses from the furnace much like a boiler economiser) is blasted into the bottom of the furnace through several tuyeres.
The coke is used as a fuel to produce the high temperatures required by the process of smelting the ore in a blast furnace. It produces CO which adds heat, as well as acting as a reducer removing the oxygen content from the ore.
Limestone is used as a flux to separate the gangue from the molten iron ore, the accumulated slag and the molten iron being tapped from two tap-holes at the bottom of the furnace. The slag goes to a disposal area and the molten iron is directed into molds known as pigs where it solidifies to pig iron and is transferred to the next stage of processing.
Converting Pig Iron to Wrought Iron and Steel.
The pig iron can be further refined to produce steel or wrought iron. Both these methods reduce the carbon content of the pig iron, which in turn reduces the brittleness property of the metals.
Wrought Iron Process
Wrought iron is produced by pummeling the pig iron using mechanically driven hammers in a wrought iron works. This process prepares it for the next stage which is the heating and forging to the desired wrought iron designs and shapes.
This used to be an art of the blacksmith, but is now normally carried out by machines.
Steel Process
This process is used to remove impurities from the pig iron produced in a blast furnace. It has developed from the early methods used such as an open hearth, or a Bessemer furnace. Today we normally use an Electric Arc Furnace (EAF) or a Basic Oxygen Furnace. (EAF)
Both methods remove a high percentage of the carbon content from the pig iron, along with the impurities of aluminum, calcium (produced during tapping), sulphur, silicon, manganese, and phosphorus.
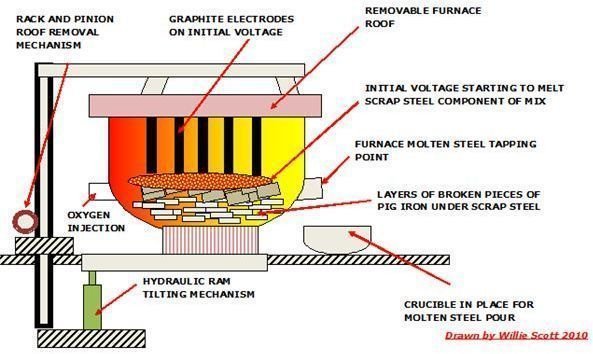
- Electric Arc Furnace (EAF)
The vertical circular steel furnace is brick-lined and has a roof which contains graphite electrodes. The electrodes are withdrawn and the roof is lifted to facilitate the loading of the pig iron to the bottom of the furnace. This is followed by a known quantity by scrap steel, which is dependent on the final grade of steel required.
Once the roof is replaced, the electrodes are lowered and an initial voltage is applied to them, and when the scrap steel has started to melt, the main voltage is applied.
When the metal is molten, oxygen is injected into it, with the impurities being converted to oxides and forming a slag. The oxygen also reacts with the carbon forming CO which is combusted adding heat to the process. Oxygen injection also reduces the carbon, nitrogen and hydrogen content on completion of the process, samples are taken and analyzed, and if acceptable, steel with content of up to 1% of carbon is poured into ingots.
- Basic Oxygen Furnace (BOF)
If the furnace has been designed to be linked to the blast furnace process, molten iron is carried in crucibles and poured into the BOF. This is a much more efficient method of removing the bulk of carbon and impurities rather than using solid pig iron
The crucible of molten iron can undergo pretreatment by the injection of magnesium, iron oxide to remove the sulphur, phosphorus, and silicon, or these impurities can be removed during the BOF process.
The BOF furnace is filled up to 20% scrap steel, and then the molten iron is poured into the furnace from the crucible.
Oxygen is injected through a water cooled steel lance into the molten metal promoting the same effects as in the EAF steel processing system.
Iron and Steel Smelting Process Sketches.
Internet Sites Visited
https://www.chemguide.co.uk/inorganic/extraction/iron.html