The Postulates of the Kinetic Theory of Gases Part Two
Collisions with Each Other
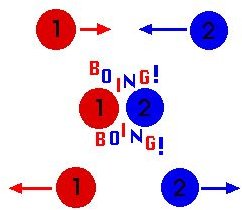
While in motion, molecules in a gas will collide
with each other, but these collisions will be elastic in nature. To recall from basic physics, this means that both kinetic energy and momentum are conserved. In other words, the molecules will smash into each other and bounce away, instead of sticking together, much like billiards in a pool table.
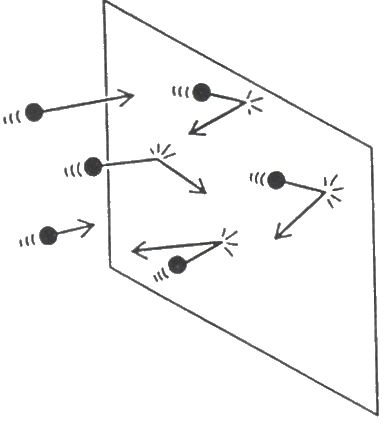
An example of an inelastic collision is presented in figure two.

Collisions within Containers
The collisions of the molecules
within container walls are also elastic.
Intermolecular Forces
The molecules don’t exert spooky-action-at-a-distance forces on each other; their potential energy consists of weakly attractive short-range forces that are electrical in nature. Their direct interactions are confined to collisions that are very short in time compared to the time between the collisions. Since the kinetic energy of these interactions is so much larger than the short-range forces due to potential energy, we can effectively neglect the latter. We therefore say that the molecules don’t have intermolecular potential energy, only kinetic energy.
Boyle’s Law Revisited
Our assumptions now allow us to gain a deeper understanding of Boyle’s Law. To recall, Boyle’s Law states that for an ideal gas, when the pressure is increased, the volume will also decrease. Conversely, if the volume is increased, then the pressure will decrease.
A container that has gas in it has its walls constantly bombarded by the molecules inside. This unceasing bombardment exerts pressure on the walls - remember that pressure is defined as the force per unit area. If you reduce the volume of gas in the container, the molecules will move closer together and strike the walls at a more frequent rate. For example, if you reduce the volume by one half, the pressure will double. Twice as many molecules will hit a given area of the walls per second. This of course is Boyle’s Law!
References
Physics for Scientists and Engineers by Douglas Giancoli
Physics For Scientists and Engineers by Raymond Serway
Essential University Physics by Richard Wolfson
Image Credits
Boing.gif from Eastern Illinois University Physics Department
Inelastic collision from Kevin Rosseel
Resources
Bright Hub Article on Molecular Internal Energy