Helium Gas: Liquefaction of Helium. Applications of Helium.
Introduction
In our previous article we learnt about the process of liquefaction of gases and the principles uses therein such as the Joule’s Thomson Effect and the use of the inversion temperature, as well as studied the arrangement for converting gaseous Hydrogen into liquid state. In this article we will take a look at a very similar procedure but for a different gas namely Helium.
The Liquefaction of Helium
You might think that there is hardly any difference between liquefaction of Hydrogen and that of Helium and probably it is quite similar at the surface but there are a few inherent differences which differentiate them.
Helium is the only substance which means fluid at temperatures below -259 degrees Celsius and its inversion point is much lower than that of Hydrogen namely around -233 degrees Celsius. The boiling point of Helium is just around -267 degrees Celsius which is quite close to absolute zero on the Kelvin scale. Just for historic reference Helium was discovered more than a century later after discovery of Hydrogen, 129 years to be precise.
The reason for more difficulty in liquefaction of Helium is that it is a noble gas which in turn means that the inter-atomic forces are quite weak and it has a low atomic mass, thereby bringing it closer to the properties of an ideal gas vis-à-vis other gases. And we know that Joule’s Thomson effect is less pronounced on gases which are close to being ideal (though no gas is fully ideal).
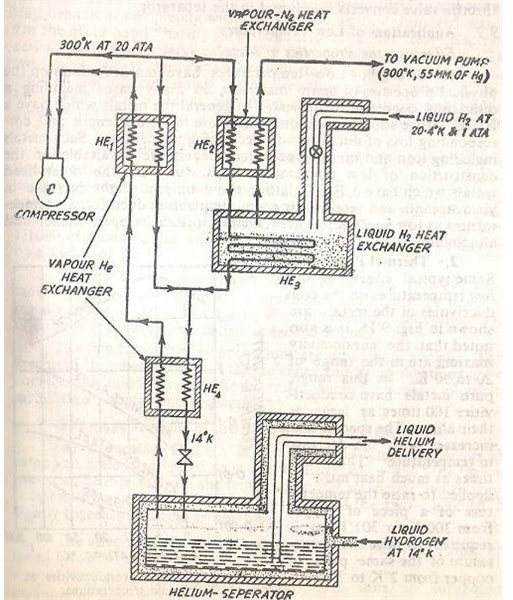
Given below is the process diagram for production of liquid Helium. The set up may seem bit similar to the arrangement of producing liquid Hydrogen studied earlier but you will note the substantial difference in the temperature and pressure readings along various points of the process.
- Initially Helium is compressed to a pressure of 20 atmospheres which raises its temperature to the region of 300 degrees Kelvin. This compressed high temperature Helium is then split into two paths.
-
The first is cooled in the heat exchanger labeled HE1 with the help of Helium vapours while the other part passes through the heat exchanger HE2 to be cooled with Hydrogen vapours.
-
Both these streams combine to be passed through the liquid Hydrogen heat exchanger HE3 before finally again getting cooled in HE4 by Helium vapours.
-
Finally the throttle valve is used to initiate the Joule Thomson effect and Helium is collected in the liquid state in the Helium separator.
Properties and Uses
Though the process described in the above section is generic in nature, in actual practice there are two main isotopes of Helium used for liquefaction namely H4 and H3 and there is a slight difference between the properties of the two in terms of their boiling point, critical temperature and so forth. The properties talked about earlier are those of H4 while the boiling point of H3 is even one degree lower.
Liquid Helium is used extensively for use in superconducting magnets which need to be cooled to extremely low temperatures during their use, that in turn are used in several fields such as say for Magnetic Resonance Imaging and Nuclear Magnetic Resonance.
References
Image of Joule Thomson Helium Liquifier - Arora, S.C. & Domkundwar, S. (1993_). A Course In Refrigeration & Air-Condioning._ Delhi: Dhanpat Rai & Sons